If you’re working in discovery, translational, and clinical development of immunotherapies, you’ve almost certainly encountered flow cytometry — in a protocol, a dataset, or a conversation with a researcher, scientist, or perhaps even the FDA. It’s one of the most established and widely used assays in the immunologist’s toolkit, and one of the standard methods for understanding the immune system at the cellular level. Yet what it actually does, how it works, and why it’s become so central to immunotherapy and clinical trials isn’t always well understood outside of the people who run it day to day.
Preface. This is a primer. Not a textbook, not a methods paper, just a clear walkthrough of flow cytometry for anyone in the biotech and pharma world who wants to understand what this assay delivers and why it matters.
What flow cytometry actually does
At its core, flow cytometry is an assay used to survey and quantify the different immune cells within the immune system. It measures what proteins are present on or inside individual cells, and it does this at a scale of thousands to millions of cells per sample, one cell at a time.
The output is a detailed profile of what immune cells are present, how frequent they are, and what functional state they’re in.
T cells, B cells, natural killer cells, monocytes, dendritic cells, granulocytes. Flow cytometry identifies them all. And beyond just naming cell types, it can distinguish functional subsets within those populations: cells that are activated versus exhausted, proliferating versus quiescent, cytotoxic versus regulatory. These distinctions matter enormously when you’re trying to understand what the immune system is actually doing in a patient.
How it works
The basic mechanics are elegant.
Flow cytometry takes advantage of antibodies conjugated to fluorescent labels, called fluorophores. Each antibody is designed to bind to a specific protein on or inside an immune cell. Those proteins act as a kind of ‘biological barcode’: a CD4+ T cell carries a different set of markers than a CD8+ T cell, which looks different from a natural killer cell, which looks different from a monocyte. By quantify cells with specific combinations of antibodies, you can read those barcodes and identify exactly which immune cells you’re looking at.
The sample, cells in suspension, is then passed through a laser-based instrument, one cell at a time, in a fluid stream. As each cell crosses the laser, the fluorescent labels emit light at characteristic wavelengths. Detectors capture that light, and the instrument records which markers are present on each individual cell and at what intensity.
The result is a high-dimensional dataset in the form of an .FCS file (Flow Cytometry Standard). For every single cell in the sample, you know which proteins were detected and how much of each was expressed. From there, you can identify cell populations through a process called gating, where two-dimensional bi-axial plots are used to distinguish cells based on their presence or absence of markers, two markers at a time (image below). This process quantifies immune cell frequencies and assesses their functional states. Sets of surface and intracellular markers tell you what kind of cell it is. Functional markers (like Granzyme B, CD69, and PD-1) reveal the state of the cell and what it’s doing. Signaling markers, like phosphorylation states, show what pathways are active most acutely. And you can query all of this simultaneously: modern cytometry assays using mass cytometry or full-spectral flow cytometry measure 40 to 50 or more markers at once, with every marker measured on every cell.
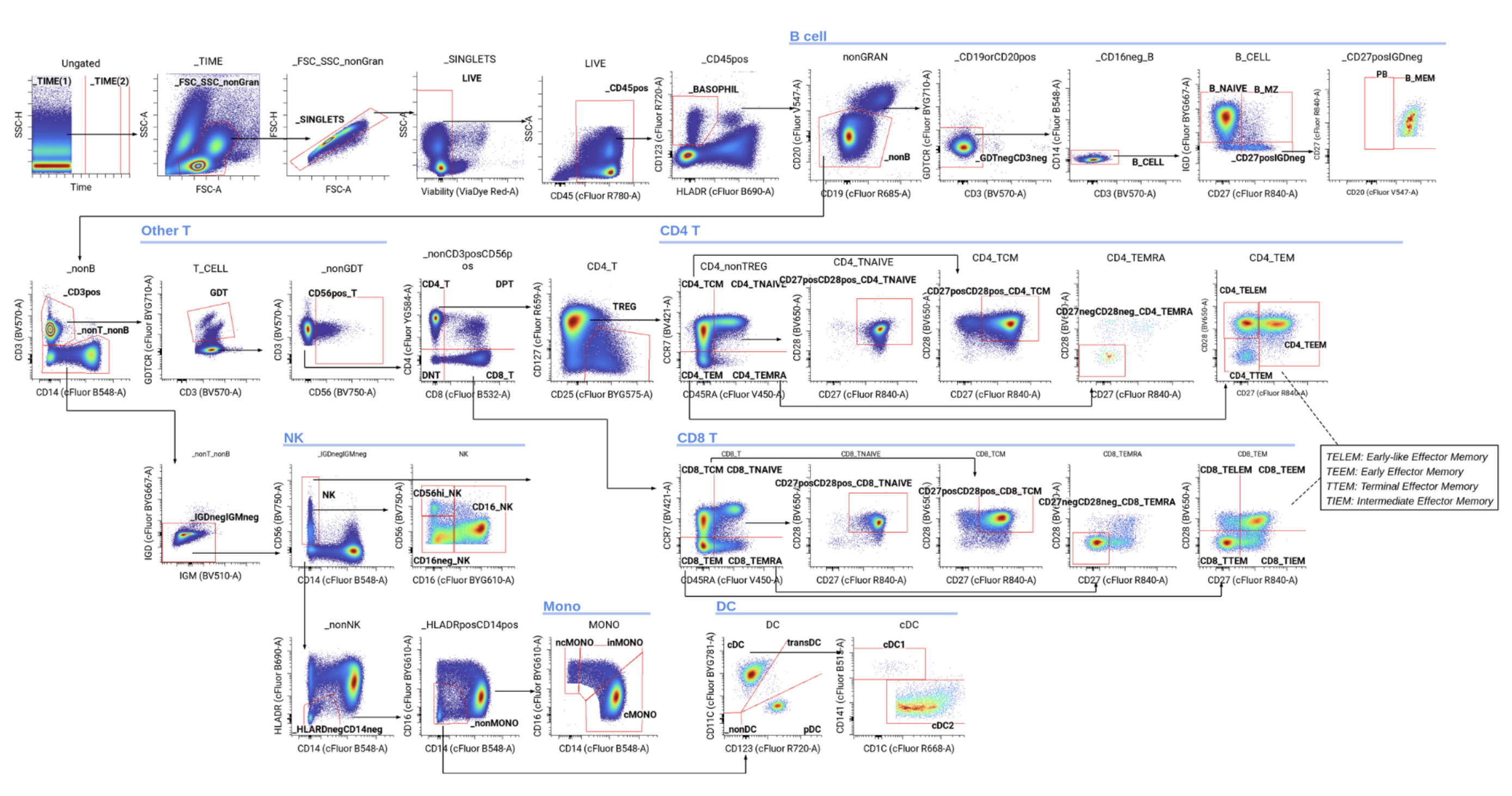
What samples can be used
One of the practical strengths of flow cytometry is its flexibility with sample types.
In clinical trials, the most common sample is peripheral blood or PBMCs (peripheral blood mononuclear cells). These are the workhorses of clinical cytometry because blood draws are relatively simple and can be collected longitudinally across treatment timepoints and measure disease states.
But cytometry is by no means limited to blood.
Bone marrow aspirates are commonly assayed, particularly in hematological malignancies like multiple myeloma. Tissue homogenates, including dissociated tumor tissue, can be analyzed when you need to understand the immune infiltrate at the site of disease. Bronchoalveolar lavage, cerebrospinal fluid, and other biofluids are also amenable to cytometry. In preclinical research, animal model samples including blood, spleen, and lymph node homogenates are routine. Anywhere you can get cells into suspension, you can run cytometry on them.
Why protein-level matters
This is one of the most important and sometimes under appreciated aspects of flow cytometry: it operates at the protein level. And proteins are what’s closest to actual biology happening in the body.
Think about it in terms of the central dogma. DNA tells you what a cell could potentially do but the presence of a gene doesn’t mean that gene is active. RNA gets you closer: it tells you what genes are being transcribed, what the cell is preparing to do. But there are still many mechanisms that modulate the journey from mRNA to functional protein: post-transcriptional regulation, translation efficiency, protein stability, post-translational modifications. Really, until you get to protein expression, you’re not seeing the actual biological output.
Proteins and enzymes are what drive cellular processes and function. They’re the effector molecules that carry out immune responses. They’re the checkpoints that regulate whether an immune cell activates or stands down. Flow cytometry reads that layer directly: not a proxy, not a prediction, not a varying level of likelihood, but the actual functional output of the cell.
Core advantages
Single cell resolution: every cell counted, every cell assayed. Every measurement is made on an individual cell, not an average across a population. This means you can detect rare subsets, identify heterogeneity within a population, and track how specific cell types respond to treatment, even when they represent a tiny fraction of the total sample.
Highly-multiplexed: 40 to 50+ markers tested per sample. Modern cytometry panels can query 40 to 50 or more markers simultaneously, with every marker measured on every cell. That means you’re not just asking “is this a T cell?” You’re asking what kind of T cell, what activation state, what exhaustion profile, what signaling pathways are active, and how that relates to the B cells, NK cells, and monocytes, all from the same tube.
Quantitative: frequencies and cell counts. Flow cytometry gives you both relative frequencies (what percentage of cells are CD8+ T cells) and absolute cell counts (how many CD8+ T cells per microliter of blood). Both are critical in clinical settings where you need to track changes over time.
Fast: completed in seconds to minutes. Once samples are stained and prepared, they can be pushed through instruments in a matter of seconds to minutes. You get readouts immediately (you literally see them on the screen of the instrument) and can start analyzing data right away. In a clinical trial, this speed translates to faster turnaround on results influencing patient recruitment, tracking disease state, and serving as critical primary and secondary endpoints.
Surface and intracellular readouts. You’re not limited to what’s on the cell surface. Intracellular staining reveals cytokine production, transcription factor expression, and proliferation markers. Phospho-flow captures real-time signaling pathway activation. You can look at the outside and inside of the cell in the same experiment.
Established and validated in the field. Flow cytometry has been around since the 1970s and has become a strong workhorse in immunology. It operates within a well-established regulatory and clinical framework, which matters enormously when you’re generating data intended to support regulatory submissions. It’s also established in the doctor's office, used to support the diagnosis of hematological malignancies and inflammatory diseases.
How it’s used in immunotherapy clinical trials
In immunotherapy trials, cytometry is a primary tool for understanding how a drug is engaging the immune system and how those immune changes correlate with patient outcomes.
At the most fundamental level, flow cytometry lets you characterize mechanism of action. Whether the therapy is a CAR-T cell product, a bispecific antibody, a T cell engager, a small molecule immune modulator, or a checkpoint inhibitor, all of these work by modulating the immune system, and cytometry is how you measure that modulation. Is the drug engaging its target? Are the expected immune populations expanding or diminishing? Is the therapy shifting cells from an exhausted to an activated state?
Beyond mechanism of action, cytometry supports target engagement studies, pharmacodynamic readouts, and biomarker discovery. You can characterize the immune landscape prior to treatment, track early on-treatment changes, and assess how shifts in immune cell frequencies and functional states correlate with clinical response. Which patients have pre-existing immune signatures that predict response? What early immune changes distinguish responders from non-responders?
Critically, the immune system never works in silos. Affecting one compartment, say, activating CD8+ T cells, inevitably impacts other immune cells: regulatory T cells, NK cells, myeloid populations. Flow cytometry’s multiplexed, systems-level view lets you see those interconnected effects rather than looking at the immune system through a keyhole.
These aren’t abstract questions. They drive go/no-go decisions, dose selection, patient recruitment, and ultimately whether a drug makes it to approval.
Three types of flow cytometry
Not all cytometry is the same. There are three main platforms, and each brings different strengths depending on the application.

Conventional flow cytometry is the most widely used and longest-established platform. It uses fluorophore-conjugated antibodies and optical detectors with bandpass filters to capture the emission peaks of each fluorophore. Instruments like the BD Fortessa and Thermo Fisher Attune are standard in both clinical and research labs. Conventional flow typically handles panels of up to 10 to 15 markers, and for most clinical trial applications, especially with well-defined panels targeting specific immune subsets, it’s sufficient. It also has the deepest regulatory track record, which matters when data needs to support submissions.
Spectral flow cytometry is a newer evolution that’s rapidly gaining traction. Instead of using discrete bandpass filters, spectral instruments capture the full emission spectrum of each fluorophore and use mathematical ‘unmixing’ algorithms to separate overlapping signals. This approach allows significantly higher-parameter panels. 30 or more markers per tube is achievable without the compensation headaches (although there are still challenges with unmixing) that come with pushing conventional instruments to their limits. The Cytek Aurora, Northern Lights, and BD SE Symphony platforms are the primary spectral instruments in the field. Spectral cytometry is especially valuable when you want deep immune phenotyping from limited sample volumes, which is often the reality in clinical trials.
Mass cytometry (CyTOF) takes a fundamentally different approach. Instead of fluorescent labels, it uses heavy metal isotope–tagged antibodies and a time-of-flight (TOF) mass spectrometer to detect them. Because metal isotopes have virtually no signal overlap, panels of 40 to 50 or more markers are achievable with minimal interference between channels. Instruments like the Standard BioTools (formerly Fluidigm) Helios and CyTOF XT are the standard platforms. Mass cytometry is particularly well-suited for deep immunophenotyping where maximum parameter depth is absolutely critical to understand a drug’s mechanism of action, and to cast the widest net for discovering biomarkers that predict patient outcomes.
Each platform has its place, and the right choice depends on the trial design, the questions being asked, and the practical constraints of sample volume, turnaround time, and regulatory requirements.
The bottom line
Flow cytometry is one of the most powerful and versatile tools available for understanding the immune system at the single-cell level. It’s been refined over decades, it operates at the protein level where biology actually happens, and it scales from basic research to large, multi-site clinical trials. It’s timely, quantitative, multiplexed, and biologically direct.
If you’re building a trial that involves immunology endpoints (mechanism of action, target engagement, biomarker discovery, pharmacodynamics), flow cytometry is very likely already part of the picture. Understanding what flow cytometry does, what it requires, and how it provides rich information on not just a part, but the whole immune system, is the first step toward getting it right.
.png)

.png)
.png)


