
In line items of a contract with your CRO, you can usually find something like a Project Management fee. What this fee exactly covers is often a little opaque. In this post, I will talk about our experience working with clients running global clinical trials, and what the Project Management fee should actually get you.
Before working with clinical trial samples, I frankly did not appreciate how important an attentive mind is to the success of a project. I was more in the camp of “if people know what they are doing, project management is obsolete”.
By now, however, I have learned about the complexities of global trials, how difficult it can be to move samples across countries, and how quickly timelines can get derailed if sight of the big picture gets lost.
To make sure we are setting projects (and our clients, and ultimately ourselves) up for success, we run every clinical trial project through 4 defined phases: Alignment, Panel Development, Clinical Processing, and Data Insights/Consultation. Each phase has defined checkpoints, structured feedback windows, and documented handoffs so there is no ambiguity about where your project stands.
Here is what that workflow looks like in practice:
Phase 1: Alignment and Initiation
Every project starts with a Kickoff Call. All stakeholders join to finalize technical specs: sample types, expected volumes, shipping logistics, metadata requirements, and panel design. The goal is to make sure we have all the information we need to handle samples from day one.
Agenda covered in every Kickoff Call:
- Sample types and expected volumes
- Shipping logistics and site coordination
- Metadata requirements
- Panel design considerations
- Timeline and Deliverables

Phase 2: Panel Development and Real-Time Updates
Once reagents and samples are in, we move into panel verification, confirming the antibody panel is performing as expected and that the gating strategy captures the cell populations you care about.
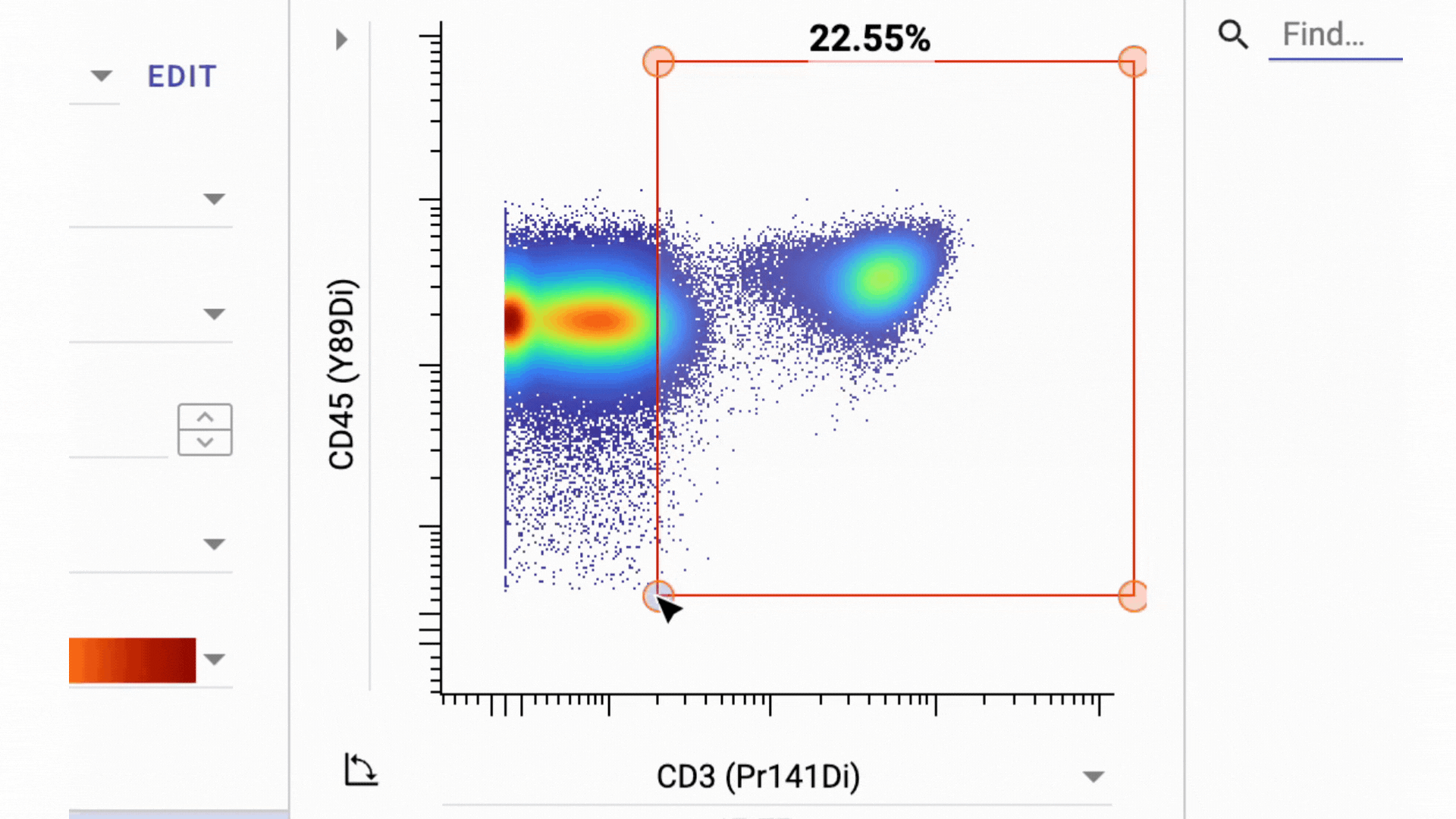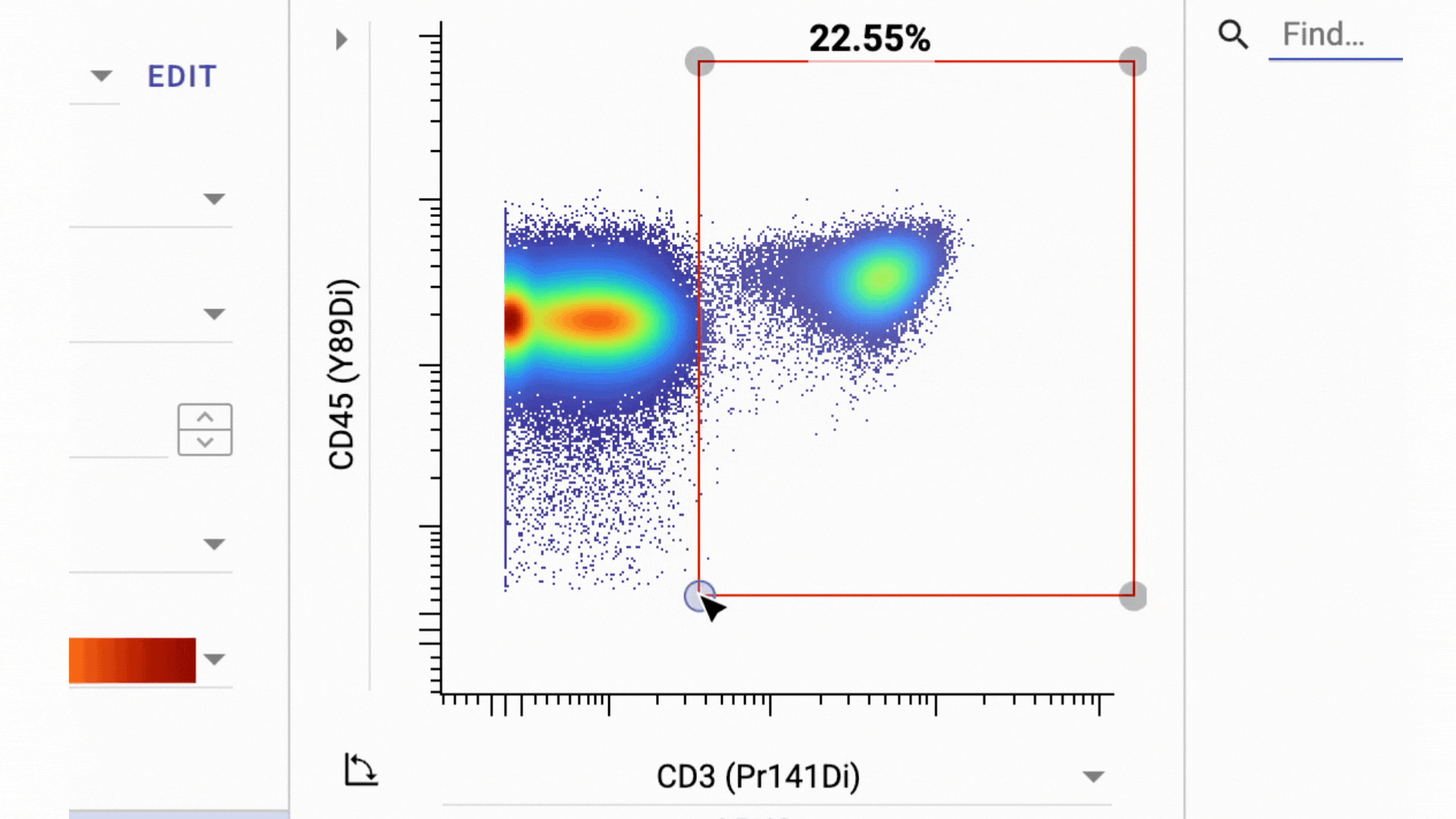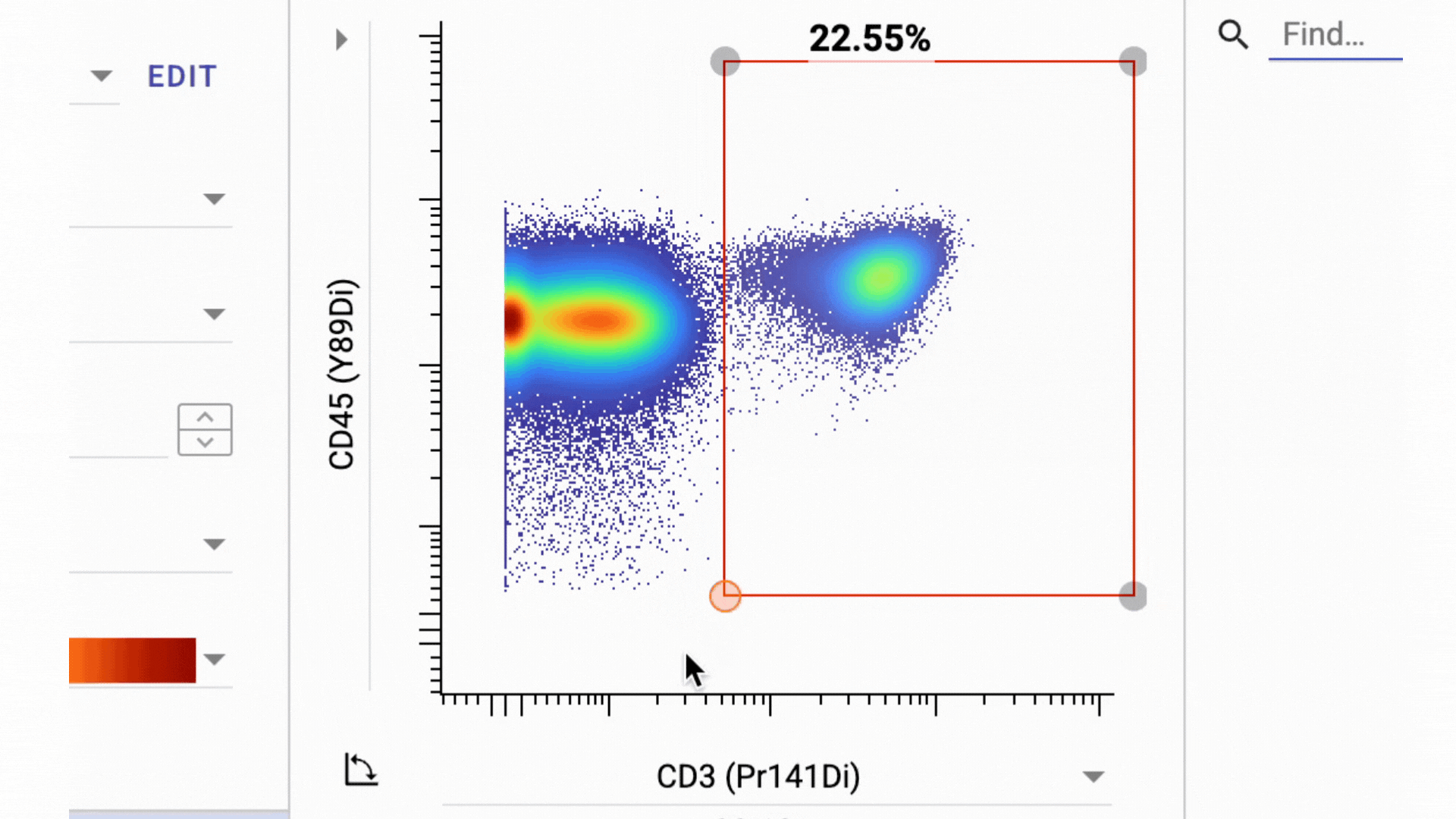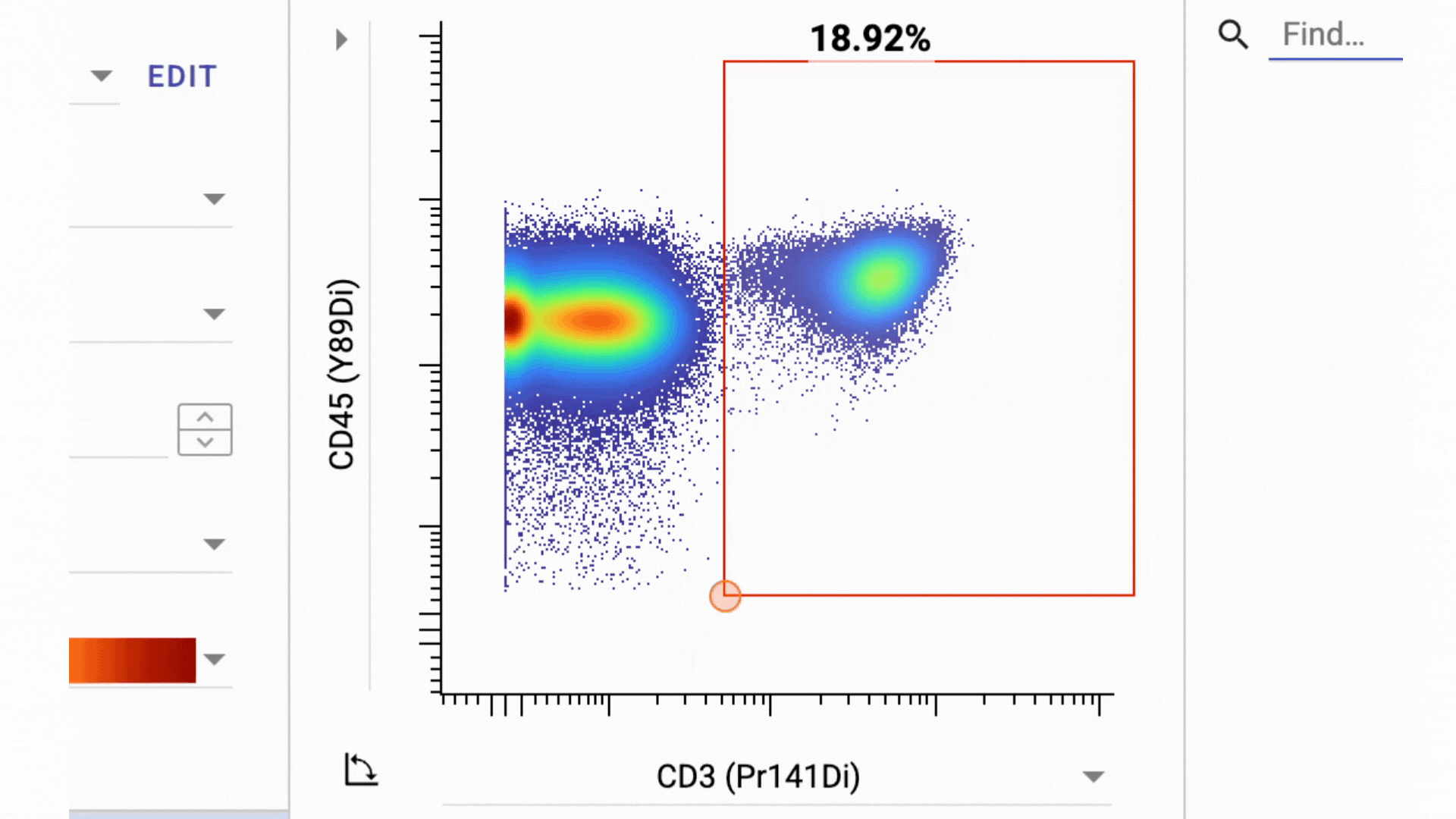
3 Business Day Feedback Window: After panel verification, we share the full gating strategy and open a 3-business-day review period. You can ask questions and request changes before we move into clinical processing.
This has actually turned into one of our most valuable features. By aligning on the gating strategy before sample processing, everyone involved knows exactly what data to expect from their samples.
We’ve heard many times that in the CRO world, samples get processed and reported in a very human-unfriendly PDF with gating strategies that are either unreadable or extremely hard to follow.
Phase 3: Clinical Processing and Quality Control
After panel verification and approval of the gating scheme, we move into clinical sample processing. This is where transparency and attention to detail becomes especially critical, because we know these samples are almost always irreplaceable.
Here’s what to expect during this phase:
- QC Checkpoints: Issues are flagged immediately, not reported in an email summary days later. If a sample doesn't meet spec, you hear about it in time to supply additional aliquots.
- Data availability Notifications: Upon completion of processing samples, notifications alert you the moment the data is live.
- Final Gate Tailoring: Clients have a second 3-business-days window to review per-sample gating directly in the Teiko dashboard, not in a static PDF. The dashboard interface lets you adjust gates and confirm results interactively before final sign-off.
Phase 4: Data Insights and Consultation
After final gate approval, we host a walkthrough call with your team. We navigate the Teiko dashboard together, highlight statistically significant findings in the context of your trial's study questions, and make sure you leave with actionable next steps, not just a data package.

Apart from our workflow, my favourite thing about project management at Teiko is that we have co-located our project management with our lab operations. This means that our project managers are intimately aware of what’s happening in the lab, and with that, where each project stands.
What this means for your trial
Every phase runs on a defined output, a defined owner, 2 structured 3-business-days feedback windows, and immediate QC reporting, from kickoff to data delivery. This ensures that you know ahead of time how your data will be structured and what to expect from it, rather than being left with a rigid, 30-page-long PDF report. If project management rigor is a criteria in selecting a CRO for your next trial, we’d happily walk you through exactly how this works in practice.
.png)
.png)




