The Contract Research Organization (CRO) model has looked similar for decades. LabCorp, IQVIA, PPD, Medpace — these companies offer a full suite of services, from site management to sample analysis. The assumption has always been that every assay fits into this model. And for many, it does. The infrastructure is built, the workflows are proven, and the data gets delivered.
But for some assays (cough, cytometry), that model isn’t working.
The cracks are showing
Cytometry is one of the most critical assays in immunotherapy trials. It drives pharmacokinetic and pharmacodynamic endpoints, mechanism of action studies, and biomarker discovery. And yet, it’s consistently one of the assays sponsors tell us their CRO doesn’t do well.
The problem isn’t that CROs are bad at what they do. It’s that we’ve been treating highly specialized assays the same as more straightforward ones. Cytometry doesn’t just require more attention to get right. It requires innovation across the full end-to-end workflow to deliver high-quality, consistent, reliable, and timely data. That means not only getting the current process right, but having a team dedicated to solving the challenges that still exist.
What happens when that level of focus isn’t there? Some sponsors, frustrated by unreliable CRO cytometry, try to bring it in-house. But that comes with its own challenges: recruiting specialized talent, maintaining instruments, building validated workflows, and scaling across a growing trial portfolio. Others take a harder path — they walk away from cytometry entirely. They drop it from their trial design. And in doing so, they lose critical cell- and protein-level data that would explain a drug’s mechanism of action and identify the biomarkers that predict which patients are most likely to benefit from treatment.
Why cytometry demands specialization
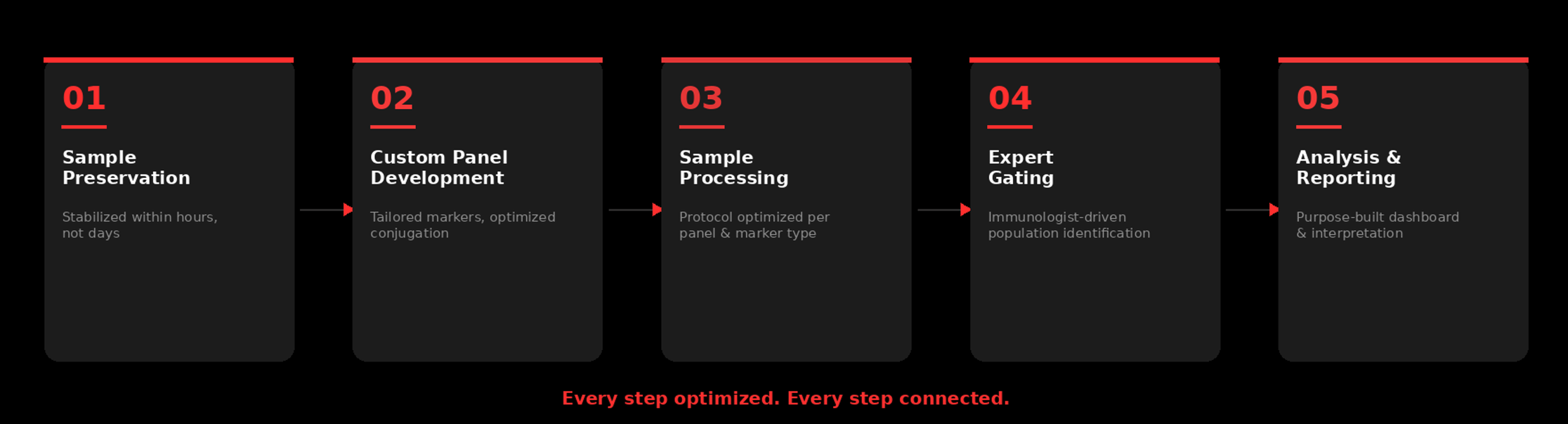
Sample collection alone is a challenge. Blood needs to be processed live within 24 to 72 hours, ideally sooner. For global trials spanning dozens of sites, getting that timing consistent is essentially unachievable. Every hour introduces variability that compounds downstream.
Panels need to be custom built for each program, with markers to quantify a drug’s target on immune cells, phosphoproteins to assess pathway engagement, activation and exhaustion markers to track CAR-T evolution. Each panel requires selecting the right metals or fluorophores per antibody to optimize detection. This isn’t off-the-shelf work.
Processing protocols have to be optimized around the panel. Surface, intracellular, and signaling proteins each have different staining requirements. Signal-to-noise needs to be maximized across the full marker set. A cookie-cutter protocol doesn’t get this right.
Gating — distinguishing immune populations and functional subsets from biaxial plots — requires immunological expertise that goes well beyond running the instrument. Building schemes, positioning gates, and doing it consistently across hundreds of samples is where deep knowledge becomes critical.
And then there’s analysis. Dose-to-dose, responders versus non-responders, single agent versus combo, treatment versus placebo — statistics, effect sizes, and the bigger picture: understanding the immune system as a system, the sum of its parts, and what the data actually means for the drug program.
Together, these make cytometry one of the most demanding assays in the clinical trial toolkit, and one that doesn’t survive the cookie-cutter approach. That’s exactly where Teiko steps in.
Michelin star, not a buffet
I sometimes think about it this way. Large CROs are the buffet — broad selection, reliable enough across the board. But for an assay as complex and high-stakes as cytometry, you need something more focused, more meticulous. You need the restaurant that does one thing and does it at the highest level.
At Teiko, cytometry is all we do. Every person, every process, every piece of infrastructure is built around making cytometry work for clinical trials. From sample preservation and blood collection in the first mile, to panel development and consistent sample processing, to analysis pipelines and a dashboard built specifically for cytometry — purpose-built for the comparisons sponsors actually need, ready to go.
That focus isn’t a limitation. It’s the whole point.
Plug & play
Here’s the thing sponsors often ask: does working with Teiko mean replacing their existing CRO? It doesn’t.
We integrate into the workflows that are already in place. Sponsors who are already working with a large CRO for their broader trial operations don’t need to change a thing. We become a piece of their existing puzzle. Our blood collection kit integrates into the larger kit system that ships to clinical sites. When samples are collected, some go to the primary CRO for their assays, and the cytometry samples come to us.
We’ve done this alongside PPD, IQVIA, LabCorp, Medpace, and CRUX. The model works because we’re not asking anyone to abandon what’s already functioning — we’re making the cytometry piece actually deliver.
The CRO model is evolving, and it’s looking specialized
The way we think about CROs and service providers for complex assays is going to change — sooner rather than later. The idea that one organization handles everything for a trial made sense when assay complexity was more uniform. But immunotherapy trials have raised the bar. The assays driving clinical decisions today require a level of specialization that the traditional model wasn’t designed to support.
The future isn’t one CRO doing everything. It’s an ecosystem of specialized partners, each doing what they do best, integrated into a single trial infrastructure. That’s where the data gets better. That’s where clinical decisions get sharper. And that’s where cytometry finally works the way it should.
.png)

.png)
.png)


