If you’ve spent time doing cytometry, you’ve heard the pushback on fixation. It masks epitopes. It over-crosslinks proteins. It makes markers harder to detect, or you lose the signal altogether. And those concerns aren’t wrong. They’re just incomplete.
Fixation done poorly absolutely distorts data.
But fixation done right is the single most important step for preserving the biology we’re actually trying to study.
What fixation does and why the balance matters
Fixation works by creating covalent bonds between proteins using a crosslinking agent. The primary mechanism involves reacting with primary amines on lysine residues to form methylene bridges, building a three-dimensional meshwork that locks proteins in place throughout the cell.
The challenge is that fixation has a narrow window when it works right. Too little time, and the crosslinking network is incomplete. Proteins leach out during washing, proteases remain active, and cells lose structural integrity.
Too much, and the matrix becomes so dense that it buries epitopes, blocks antibody binding, and distorts protein conformation. This is the experience most people reference when they say fixation doesn’t work for cytometry. And if the only options were no fixation or bad fixation, they’d be right to avoid it. Default to staining fresh sample.
But there is a sweet spot. Enough crosslinking to preserve structure, but not so much that antibodies can’t bind their targets. Modern antibody development, clone selection, conjugation chemistry, and panel validation have made this balance increasingly achievable.
The idea that fixation automatically ruins cytometry data is outdated, based on experiences with protocols that were never optimized for this purpose.
What changes when fixation is done right
When samples are fixed properly and early, within hours of collection rather than days, something huge unlocks: a preserved biological snapshot that can be shipped, stored, batched, and analyzed without the clock working against it.
That changes the entire logistics for cytometry in clinical trials.
Sites don’t need to process fresh blood on-site. Samples can ship to a central lab without degradation. Batching becomes possible. Real-time is efficient. And because the biology is locked in, the data at the end (finally) reflects what was happening in the patient at the time of collection. And that’s the point.
Compare that to the current cytometry standard: live blood shipped overnight, processed at 24+ hours, with 15% or more of the sample already degraded before an instrument ever touches it. One path captures biology (that’s what we want!). The other captures an approximation that gets worse with every passing hour (eek, lots of variability, lots of noise in the data).
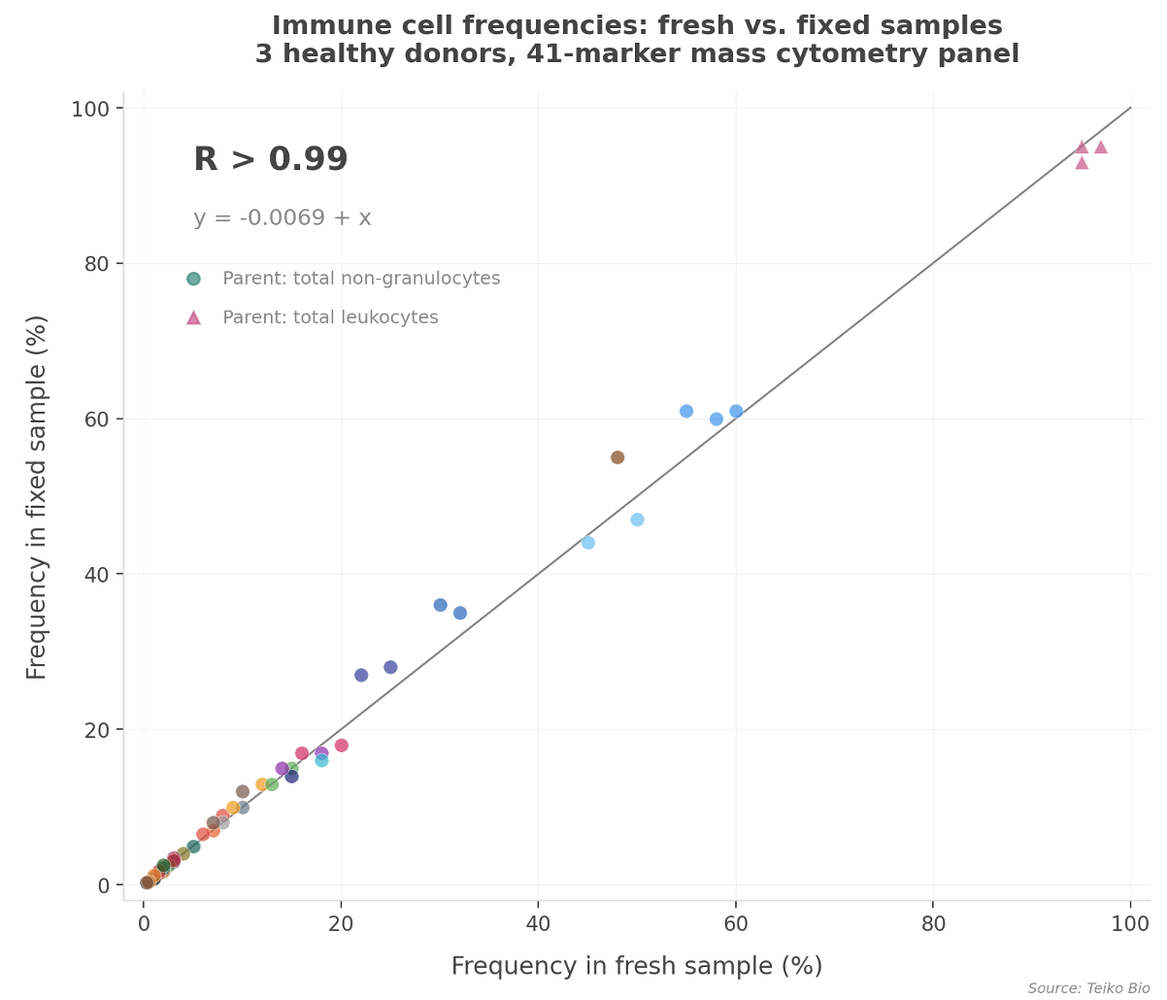
We measured immune cell frequencies in fresh and fixed samples side by side. The result: a correlation above 0.99, with the regression line nearly indistinguishable from the y = x. Fixation preserved the biology.
Other fields already figured this out
In pathology, when tissue is excised from a patient, it gets fixed in formalin and embedded in paraffin (FFPE) almost immediately. That process preserves morphology and biology for years (often decades!). Flash-freezing in genomics works very similarly. So do stabilization reagents in RNA sequencing. The goal is always the same: capture a biological snapshot before drift occurs.
So why does cytometry still rely on shipping live, unfixed blood across the country and hoping the biology holds?
The vision: immediate fixation
Right now, our approach at Teiko is 3 steps (room temp, no centrifuge – it’s easy!) stabilizes samples within approximately two hours of collection — a massive improvement over the 24–72-hour industry standard. But the gold standard we’re working toward is immediate fixation. Vein to collection tube to anticoagulant to fixation, no gap (no cap). That’s the point at which we’re truly capturing native biology. Look for an update in the near future. ;)
Rethinking the conversation — fixation for clinical trials
The skepticism around fixation is understandable. It comes from real experiences with lose of signal. But the answer isn’t to avoid fixation — it’s to do it better. Optimized crosslinking, modern antibody development, and thoughtful panel design make it possible to preserve biology and still detect it.
The status quo, continuing to ship, processing, and analyze live blood samples and accepting that the biology will drift, isn’t working. We’ve been watching it not work for decades. It’s time to fix the fixation problem, and in doing so, fix cytometry and biology.
.png)

.png)
.png)


