Every biological measurement starts with a sample. It doesn't matter how sophisticated the instrument is, how validated the panel is, or how advanced the analysis pipeline is. If the sample no longer reflects the biology being studied, then every result that follows is already compromised.
The analysis will be precise. The statistics will be clean. And the conclusions will be wrong. Not starting the race behind. Disqualified at the starting line.
This is true across biology: genomics, proteomics, single-cell workflows, tissue-based assays. But the place I see it play out the most — because of my work — is cytometry. And the gap between what the industry accepts and what the biology demands has been bothering me for years.
The cycle sponsors know too well
As an immunologist, I used cytometry in the lab. Small scale, consistent protocol, direct oversight, single operator (me!). In that environment, cytometry is incredibly powerful. But clinical trials are a completely different reality. No longer one lab with one instrument — instead, fifty, a hundred, or more global sites. And what was once precise becomes highly variable.
In conversations with drug sponsors, from emerging biotech to top-10 pharma, a consistent cycle emerges. They start with a large Contract Research Organization (CRO). They hit data quality issues: QC failures, variability, delays. So they bring it in-house. It works until the trial scales. Then the infrastructure can’t keep up, and they go back to a CRO, hoping it’ll be different.
CRO → frustration → in-house → scaling failure → back to CRO. Repeat.
This cycle isn’t poor decision-making. It’s a logistical bottleneck that neither model solves.
Everything matters, and it all compounds
Cytometry at scale has no single point of failure. It has many, and they stack on top of each other.
The operator matters — multiple technicians handling samples differently across dozens of sites. The instrument matters — different cytometers, configurations, calibration states. The gating matters — one analyst’s gates can look completely different from another’s. The analysis pipeline matters — some groups don’t even have access to proper tools. And the time blood sits before processing matters — because immune cells are living, dynamic systems that start drifting the moment blood leaves the body.
Every factor contributes. But if the starting sample no longer represents the system being studied, nothing downstream can fix that. Already running a race that can’t be finished.
The hot take: 100% of samples are biologically distorted by 24 hours
The industry standard says samples should be processed within 24 to 72 hours. But we need to be honest about where that window came from. It wasn’t set by biology. It was set by logistics because that’s how long shipping and scheduling typically take. Not because anyone demonstrated that a 24-hour-old sample still looks like what was in the patient.
And just because something is ‘protocol standard’ does not mean it is scientifically right.
These are living cells. They’re metabolizing glucose, shifting immune states, undergoing apoptosis. A sample processed at 24 hours may still generate data. But it is no longer a true snapshot of in vivo biology. Many sponsors quietly observe 20% or more sample loss due to logistics alone, plus QC failures, operator variability, and noisy immune readouts. That’s often considered normal. It shouldn’t be.
The data below — from work we presented at AACR IO 2025 — shows what happens to fresh blood samples as they sit outside the body:
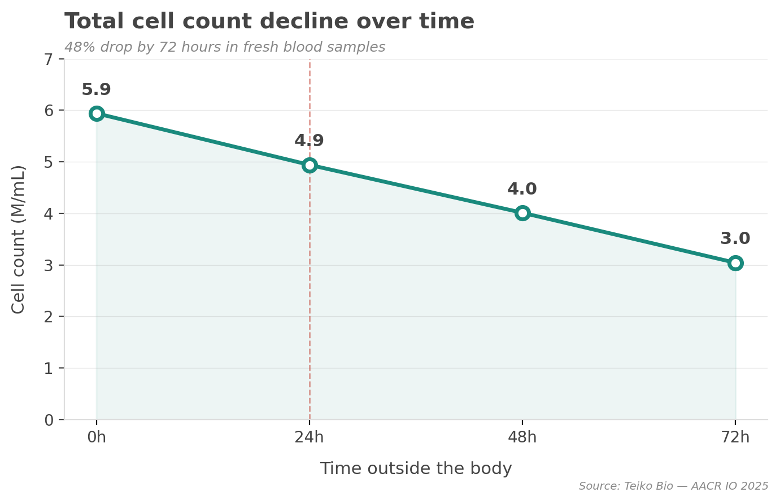
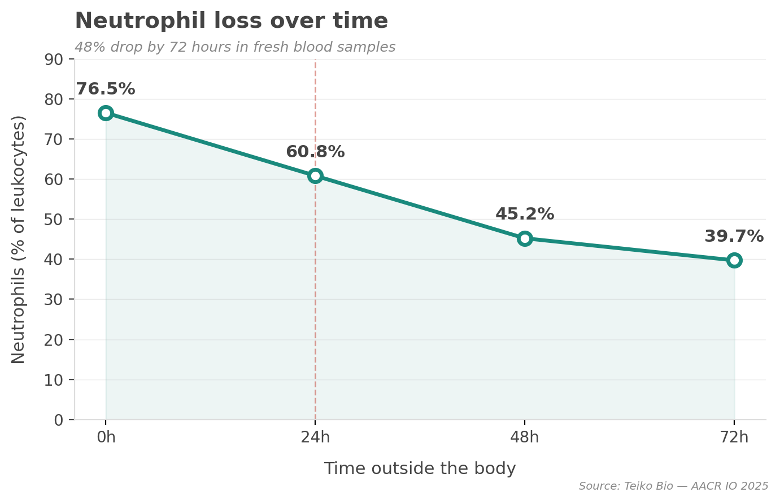
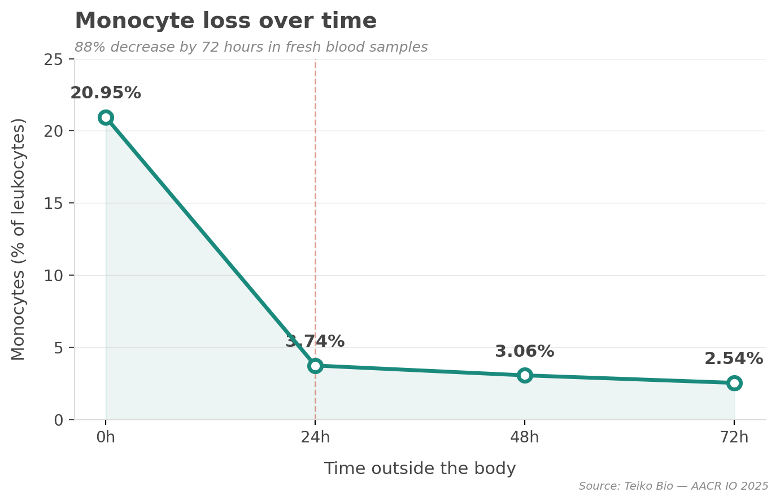
Neutrophils drop 48%. Monocytes drop 88%. Total cell count cut nearly in half by 48%. And this is within the window the industry considers acceptable.
Why this matters beyond the lab
This isn’t just a data quality problem. It’s a clinical decision problem. Sponsors use cytometry data to interpret mechanism of action, discover biomarkers, stratify patients, and define translational endpoints. If that data is distorted from the start, the wrong patients get enrolled, true responders get missed, and mechanism of action gets misread.
Consider the broader context: the overall rate of drug approvals still sits below 10%. There are many reasons for that. But when we look at the upstream inputs — the biological data that informs which drugs move forward and which patients get selected — we have to ask: how much of that failure rate is influenced by data that never accurately reflected human biology in the first place?
The real bottleneck is the first mile
When data quality issues arise in cytometry, the industry focuses on gating, panel design, instrument harmonization, and analysis pipelines. All important. But also all downstream. The true inflection point happens earlier: vein to collection to stabilization.
Instead of asking “Did we process the sample within 24–72 hours?” we should be asking “How quickly was the biology stabilized after collection?” With our approach at Teiko, samples are stabilized within approximately two hours, often within 30 minutes of the draw. The long-term gold standard — what we’re ultimately working toward — is immediate fixation. Vein to collection tube to anticoagulant to stabilization, with no gap.
Breaking the cycle
I’ve been at Teiko for over four years now, building from the ground up. Every week I’m talking to translational and biomarker leads, clinical operations teams, and drug sponsors. These conversations aren’t theoretical. They’re about real trials, real data, and real frustration. I think about these problems every day. Seriously, every day.
The CRO-to-in-house loop won’t break itself. It breaks when we solve the first-mile problem and build end-to-end infrastructure that makes clinical cytometry actually work at scale. And the principle extends beyond cytometry. If the system we’re measuring has already drifted from human biology, the answer is to close the gap. Not accept it. Not call it standard.
If the sample has drifted before stabilization, we are no longer studying the patient's biology. We're studying time outside the body. That's a bottleneck the industry can no longer afford to accept. We're fixing it.
.png)

.png)
.png)


